Notice of Intent – Proposed Amendments to the Existing Requirements of the Consumer Chemicals and Containers Regulations (CCCR, 2001)
By: Teah Jazey, BAT (HONS), REGULATORY AFFAIRS AND PRODUCT SAFETY ASSOCIATE, email
On July 11th Health Canada published a Notice of intent – Proposed new requirements for consumer chemical products under the Canada Consumer Product Safety Act. This is an update that has been rumored for quite a while – currently Health Canada is looking for feedback and comments from interested stakeholders on the proposed amendments to add Human Health Hazards of Concern (HHHNOCs) to the CCCR, 2001 set of classifications.
Read to the end to see how you can participate during this consultation period! Let our team know if you have questions. Dell Tech is here to help with any questions you may have about your consumer products and the requirements for sale in Canada.
CCCR 2001: Current Regulations
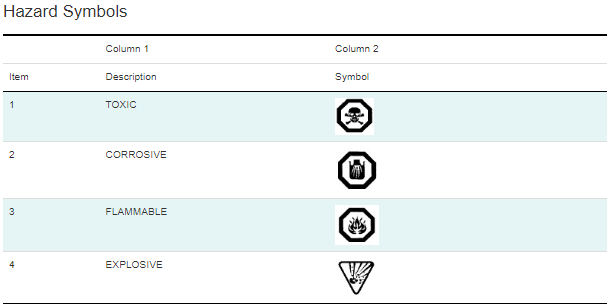
The current set of regulations for consumer products, CCCR, contains only 5 health and physical hazards:
- Acute toxicity
- Corrosive
- Flammable
- Quick Skin Bonding Adhesives
- Pressurized Containers
The following symbols are the only currently acceptable hazards symbols under CCCR:
Though a product might be considered carcinogenic or a skin sensitizer, currently it would not be classified as such. Under the current set of consumer regulations these are not acceptable classifications as they would not fall under any of the categories shown above.
Health Canada Considers Aligning CCCR with the Hazardous Product Act (HPA) and Regulations
Currently consumer products and workplace products, regulated under the Hazardous Products Act and Regulations, are not aligned. In an attempt to harmonize the two set of regulations Health Canada is considering the addition of HHHNOCs to be added to the CCCR, 2001.
Human Health Hazards of Concern or HHHNOCs have longer term effects on humans and can include:
- Carcinogenicity
- Reproductive Toxicity
- Respiratory or Skin Sensitization
- Germ Cell Mutagenicity
- Specific Target Organ Toxicity (single or repeated exposure)
The CCCR, 2001 was originally implemented as a way to protect Canadian consumers. The potential addition of these HHHNOCs is an extension of this and the goal is for Canadians to be aware of the hazards they may be exposed to when using products.
Upon stakeholder feedback Health Canada will consider fully replacing the CCCR, 2001 with a risk-based GHS framework for product classifications and hazard information disclosures. This would be considered the alternative regulatory approach to the proposed amendments in this Notice of Intent.
Health Canada Needs Your Feedback
The Government is looking for feedback and comments from manufacturers, importers, retailers, industry associations and the general public about these proposed changes. The below link contains suggested questions by Health Canada – these questions can be found at the bottom of the notice. Some of the questions are:
- Do you agree that the proposed classification criteria and information disclosure requirements for HHHOCs (i.e., carcinogenicity, germ cell mutagenicity, reproductive toxicity, specific target organ toxicity, and respiratory or skin sensitization) in consumer chemical products based on those set out in the Globally Harmonized System (GHS) of Classification and Labelling of Chemicals (ST/SG/AC.10/30Rev.8) would be reasonable and effective?
- If you do not support the proposal outlined in the NOI, do you support the alternative regulatory approach outlined in the NOI? The alternative regulatory approach proposes to fully replace the CCCR, 2001 with a risk-based GHS framework for classification and information disclosure and mandating additional protections (e.g., prohibitions, restrictions and child-resistant containers).
- Do you support the introduction of additional protections, such as prohibitions, restrictions, or child-resistant container requirements, for certain HHHOCs in consumer chemical products?
If you are interested in participating in this consultation you will need to submit your comments to ccpsa-lcspc@hc-sc.gc.ca. You will need to include a contact name, organization name and contact email. The consultation period for this notice will be effective for 90 days, from July 11 2023 to October 09 2023.
For more information, the Notice of Intent can be found here.
CCCR 2001 and Health Canada Recalls
Did you know that Health Canada can recall your product for non-compliance with CCCR? Even if your company is not located in Canada, if you sell in this country, Health Canada has the right to remove your products from the shelf or even stop shipment at the border.
Recall is an expensive, time-consuming process. In addition to penalties from the government and your suppliers, recall notices can end up on social media – directly available to consumers. Avoid this negative outcome by working with Dell Tech’s trusted experts to conduct a chemical label review of your products.
CCCR, 2001 under the Canada Consumer Product Safety Act (CCPSA) protects Canadians from hazards posed by chemical consumer products sold in stores or online in Canada by regulating the disclosure of all hazards on consumer product labels. The regulations stipulate labeling and packaging requirements for chemical products aimed at informing the consumer of potential hazards posed during use. Canadian consumers will find information on toxicity, flammability, corrosivity, and quick skin-bonding adhesives and if they apply to the products that they are using.
We know end-user safety is of paramount importance to you and your organization. Let Dell Tech help you keep customers safe while ensuring compliance with Canada’s Consumer Chemicals and Containers Regulations, 2001 (CCCR 2001). Our knowledgeable and professional Technical Consultants will review chemical product formulations and provide a report including a template with the bilingual precautionary text necessary for your product label.